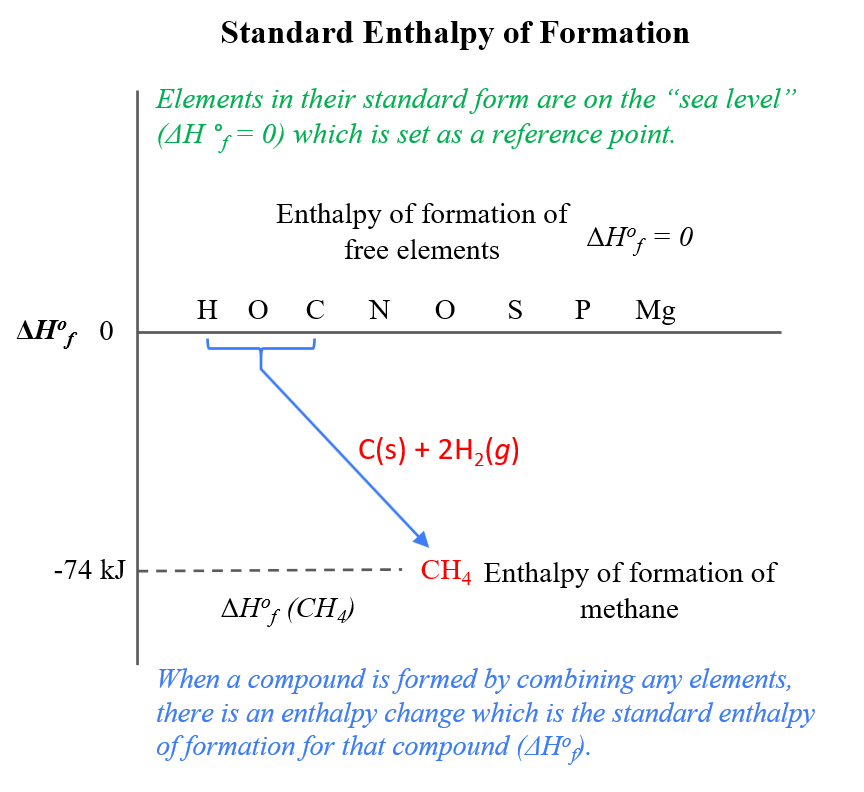
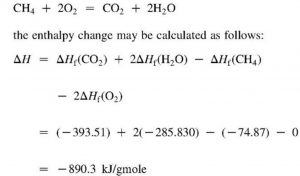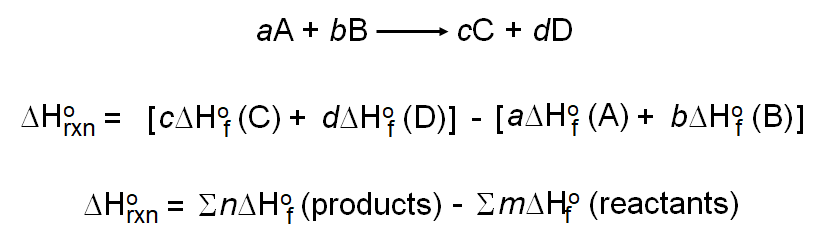Given the following equations calculate the standard enthalpy of the reaction, 2Fe(s) + 3/2 O2(g) → Fe2O3(s) H^o = ? (i) 2Al(s) + Fe2O3(s) ⟶ 2Fe(s) + Al2O3(s) H^o = - 847.6

SOLVED: The combustion of tample of aluminium poduces 025 molof aluminium oxide and feleases 419.0 kJ of heat at standard conditions. Al () + % 0z (g) ++ B ALOs () Define
The heat of combustion of C, H2 and CH4 at 298 K and 1 atm are respectively-393 kJ/mol, -286 kJ/mol, and -892 kJ/mol. How do I calculate the enthalpy of formation for

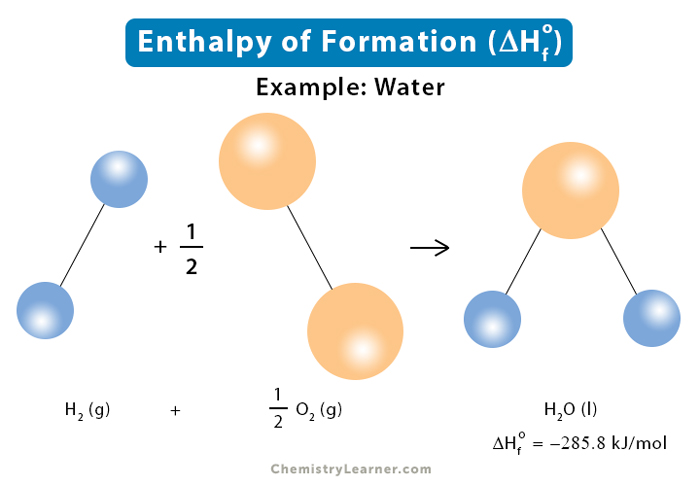
Enthalpy of combustion of carbon to CO2 is -393.5KJ mol-1. Calculate the heat released upon..... - YouTube