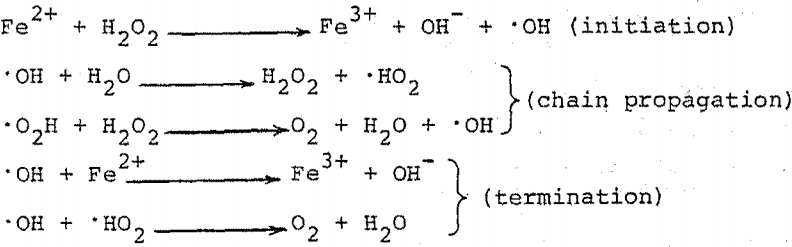Oxidation of Wine Phenolics: A Critical Evaluation and Hypotheses | American Journal of Enology and Viticulture

Objectives Define oxidation and reduction in terms of electron loss and gain. Deduce the oxidation number of an element in a compound. State the names. - ppt download

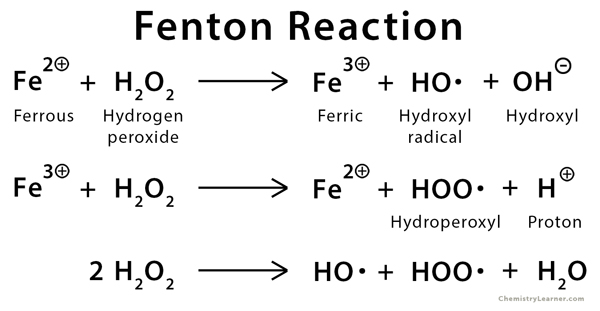
Is Sulfate Radical Really Generated from Peroxydisulfate Activated by Iron(II) for Environmental Decontamination? | Environmental Science & Technology

fenton reagent reaction hydrogen peroxide iron sulfate | Fundamental Photographs - The Art of Science

Iron Supplements and Magnesium Peroxide: An Example of a Hazardous Combination in Self‐Medication - Vrolijk - 2016 - Basic & Clinical Pharmacology & Toxicology - Wiley Online Library
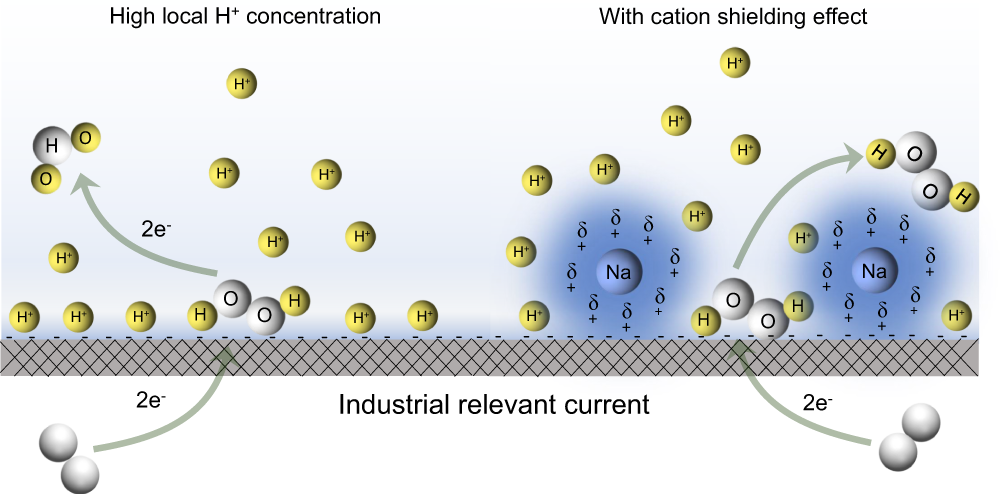
Electrochemical oxygen reduction to hydrogen peroxide at practical rates in strong acidic media | Nature Communications

Heat of reaction between ferrous ammonium sulphate and hydrogen peroxide in aqueous solution - Transactions of the Faraday Society (RSC Publishing)

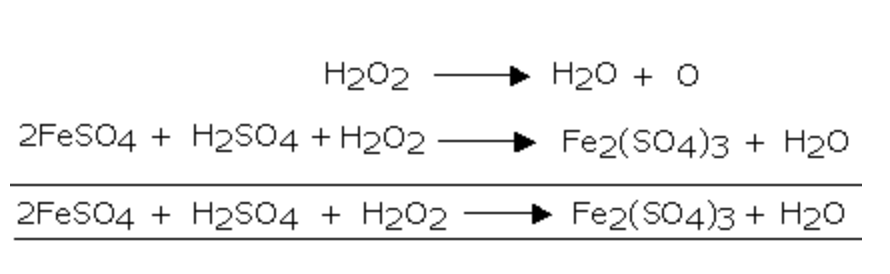
What is the reaction for oxidation of ferrous sulphate to ferric suphate by a strong oxidising agent - Chemistry - - 3772801 | Meritnation.com

fenton reagent reaction hydrogen peroxide iron sulfate | Fundamental Photographs - The Art of Science

Growing crystals of Ammonium Iron III Sulfate ( Ferric Alum). It's a double salt of Ammonium Sulfate and Iron III Sulfate. I made it by reaction of Hydrogen peroxide with Iron II

fenton reagent reaction hydrogen peroxide iron sulfate | Fundamental Photographs - The Art of Science