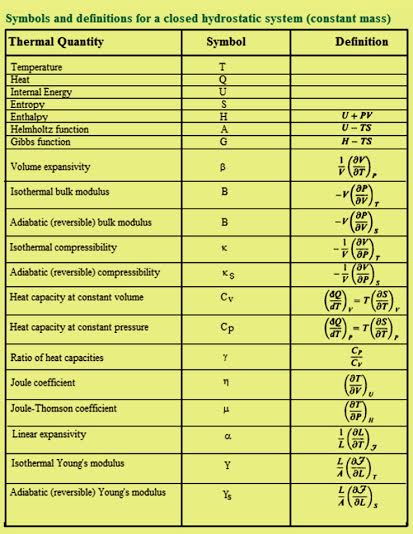
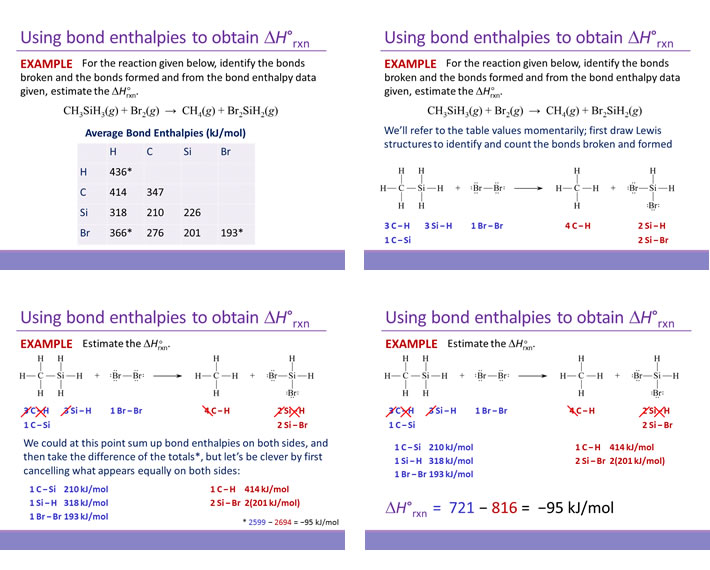
Unit Thermochemistry Thermochemical Equations Worksheet 3 Answers Form - Fill Out and Sign Printable PDF Template | signNow

SOLVED: Using the following thermochemical equations, calculate the standard enthalpy of combustion for one mole of liquid acetone (C2H6O). C2H6O (I) + 4 O2 (g) â†' 2 CO2 (g) + 3 H2O (

Given, C(graphite + O2(g) → CO2(g); ΔrH^o = - 393.5 kJ mol^-1 H2(g) + 12O2(g) → H2O(l); ΔrH^o = - 285.8 kJ mol^-1 CO2(g) + 2H2O(l) → CH4(g) + 2O2(g); Δ^rH6o = +